
Opioid Addiction Treatment Options
Developing the courage to begin a medication-assisted treatment (MAT) program for opioid addiction can be difficult – we understand. Our staff at AppleGate Recovery are non-judgmental and committed to delivering confidential, individualized care. AppleGate Recovery specializes in outpatient MAT programs for opioid use disorder (OUD). We also offer comprehensive treatment plans based on your individual needs that include buprenorphine and Suboxone alongside counseling and recovery services. We at AppleGate Recovery, help you identify what your individual recovery looks like and more importantly, how to get there.
Advantages of Suboxone
- Approved by the FDA for clinical use in 2002, buprenorphine (Suboxone) has been proven to be effective in the treatment of opioid addiction.
- The first opioid treatment medication approved for this use, buprenorphine can be prescribed by a medical provider and filled at any pharmacy offering flexibility and freedom from daily program visits.
- A long-acting medication that when dosed daily, prevents cravings and withdrawals for 24 hours or more. As patients stabilize, physicians may reduce the dosing based on your individual needs.
- Overdose on buprenorphine is unlikely if patients take the medication as prescribed, following all safety precautions and drug interaction warnings.
- Buprenorphine compounds with naloxone act as effective deterrents to misuse.
- Suboxone and Subutex are registered trademarks of Indivior UK Limited.
- Zubsolv is a licensed trademark of Orexo US, Inc;
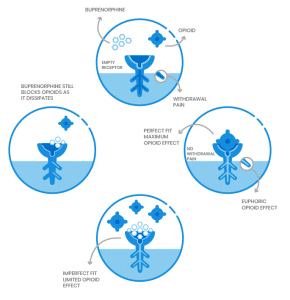
How Suboxone Treatment Works
Buprenorphine with Naloxone, commonly known as Suboxone, reduce the effects of opioid withdrawal and ease the cravings experienced due to those effects. They do this by attaching to the same receptors in the brain that other opioids do, eliminating the physical withdrawals so that patients can focus on their recovery.
Buprenorphine is a partial agonist which activates the opioid receptors in the brain, but to a much lesser extent than full agonists. It has a “ceiling effect”. This means that as you continue to take more of the medication, the effects do not continue to increase. Unlike commonly misused opioids, buprenorphine has the ability to control withdrawal symptoms without being used to “get high”. Additionally, it blocks other opioids from attaching to the receptors in the brain.
Compound medications including Suboxone and Zubsolv combine buprenorphine with naloxone. Naloxone causes no opioid effect and has no negative reaction when taken under the tongue as prescribed. However, if misused or injected it will cause immediate withdrawal symptoms. This deterrent helps to reduce misuse and diversion.
Individualized Treatment Plans

Each patient works with their medical provider and counseling staff to develop a treatment plan that addresses their unique needs. Those plans include:
- Medication-assisted treatment with buprenorphine and buprenorphine compounds including Suboxone, Bunavail & Zubsolv
- Ongoing medical and clinical assessment
- Individual counseling
- Group counseling
- Case management
- Life skills training
- Referral to community resources
- Smoking Cessation Assistance
- Lab services – including Hepatitis C and HIV testing (Select Locations)
Treatment during Pregnancy

Pregnancy is an exciting and challenging time for any mother, but those in recovery may require a higher level of care and support. AppleGate Recovery understands the importance of our role in the lives of our pregnant patients, and works to provide support during and after the pregnancy. This includes:
- Active communication with the obstetrical physician and team
- More frequent visits as the pregnancy progresses for the same monthly fee
- Assistance with tobacco cessation
- Ongoing medication assessment and adjustment as needed to support changes during pregnancy
- Discussion of the benefits of and concerns about breastfeeding for both mother and baby
- Guidance on minimizing Neonatal Abstinence Syndrome as much as possible
- Engagement in recovery during the critical post-partum period

Medication Dosage
An important part of your treatment is working with an experienced medical provider to determine the medication and dosing that is right for you. A proper dose in Suboxone treatment varies for each person. While the medications have been shown to be more effective at lower doses, most providers typically begin relatively aggressively. The ceiling effect of the medication makes this approach safe, and supports the goal of eliminating withdrawal symptoms, helping patients stabilize more quickly.

What is OBOT and is it effective?
The flexibility of outpatient office-based opioid treatment (OBOT) allows you to seek addiction treatment without putting your life on hold. This form of recovery uses an appointment system which is beneficial for those who have busy lives. Unlike methadone clinics, where you would be required to make daily visits for a dose of medication, as an OBOT patient you are given prescriptions for medications containing buprenorphine. This partial agonist can safely be self-dispensed at home due to its opioid blocking abilities and ceiling effect, meaning it will not produce a euphoric high.
Another benefit of OBOT is the ability for you to speak with your medical provider about co-occurring diagnoses regarding your mental and physical health. Medical providers understand patients with substance use disorder may have additional needs and can refer you to local health organizations to address these underlying health conditions. Addressing these underlying health disorders can significantly improve and sustain recovery in the long run. With the help of substance use counseling, you can take complete control over your treatment and accountability in the program with access to the facility’s resources and open communication with medical staff and providers.
Phases of Treatment
From the Substance Abuse and Mental Health Services Administration, SAMHSA, Suboxone treatment happens in three phases:
The Induction Phase is the medically monitored startup of Suboxone treatment performed in a qualified physician’s office or certified OTP using approved Suboxone products.
The medication is administered when a person with an opioid dependency has abstained from using opioids for 12 to 24 hours and is in the early stages of opioid withdrawal.
It is important to note that Suboxone can bring on acute withdrawal for patients who are not in the early stages of withdrawal and who have other opioids in their bloodstream.
The Stabilization Phase begins after a patient has discontinued or greatly reduced their misuse of the problem drug, no longer has cravings, and experiences few, if any, side effects.
The Suboxone dose may need to be adjusted during this phase. Because of the long-acting agent of Suboxone, once patients have been stabilized, they can sometimes switch to alternate-day dosing instead of dosing every day.
The Maintenance Phase occurs when a patient is doing well on a steady dose of Suboxone. The length of time of the maintenance phase is tailored to each patient and could be indefinite.
Once an individual is stabilized, an alternative approach would be to go into a medically supervised withdrawal, which makes the transition from a physically dependent state smoother.
People then can engage in further rehabilitation—with or without MAT—to prevent a possible relapse.



